Biological Science Faculty Member - Retired
Dr. Walter R. Tschinkel
- Office: Biology Unit I
- Area: Ecology & Evolution
- Lab: Biology Unit I
- E-mail: tschinkel@bio.fsu.edu

Robert O. Lawton Distinguished Professor of Biological Science, Emeritus
Ph.D., University of California, Berkeley, 1968
Smith, J. J., B. F. Platt, G. A. Ludvigson, and J. R. Thomasson. 2011. Ant-nest ichnofossils in honeycomb calcretes, Neogene Ogallala Formation, High Plains region of western Kansas, U.S.A., Paleogeology, Paleoclimatology, Paleoecology 308:383-394. This publication presents Daimoniobarax tschinkeli, a new species of fossil ant nest named for Dr. Walter R. Tschinkel.
The Fire Ants by Walter R. Tschinkel (Harvard University/Belknap Press, 2006)
Ant architecture: the Wonder, Beauty and Science of Underground Nests. by Walter R. Tschinkel, Princeton University Press. 227 pp, 100 illustrations.
Major Research Topics
Social control of queen fertility
Evolution of colony life histories
Population biology of fire ant colonies
Fire ant, native ant interactions
Architecture of subterranean ant nests
Natural history of arboreal ants
Chemical defense, pheromones and systematics of tenebrionid and other beetles
Inhibition of metamorphosis by crowding
Causes and characteristics of Namibian fairy circles
Essays on Education
Fall 2007 commencement address, Florida State University
Research and Professional Interests:
Ants are among the most important organisms found in mid- to low-latitude terrestrial ecosystems, often playing keystone roles. Their success is due largely to their highly social way of life; they live in colonies ranging in size from a few dozen to several hundred million individuals. In many ways, ant colonies function like superorganisms—tasks and physiological roles (including reproduction) are divided among the queen and several types and ages of workers. My research recognizes the organism-like nature of ant colonies: each colony most commonly begins life as a single individual and goes through a process of growth, development, and death; specific colony functions are carried out by specialized classes of workers, larvae, or sexual individuals; competition takes place among colonies, just as it does among individuals of nonsocial animals; nourishment must be taken in by colonies, distributed, and allocated among colony members and colony functions; colonies must produce specialized sexual individuals (propagules) for reproducing themselves, and these must leave the colony, mate, and found new colonies.
Research Areas
No Longer Active
Chemical defenses and natural history of tenebrionid beetles, including the
evolution of defensive systems, and the evolution of life history
characteristics.
Videos
Mystery Below Ground: Architecture of a Harvester Ant Nest - Video: AVI 3gb, MKV 300mb, WMV 63mb
Mystery Below Ground: Architecture of a Harvester Ant Nest
YouTube Channel: Ants and Much Much More. Videos of ant biology, wildlife, art and family history.
Annotated research areas and publications
(1) General and Review: Advocacy for more systematic collection of social insect data and emphasis on natural history and life cycle studies.
Tschinkel, W.R. (2021) Ant architecture: the Wonder, Beauty and Science of Underground Nests. Princeton University Press. 227 pp, 100 illustrations.
Tschinkel, W.R. and E.O. Wilson (2014). Scientific Natural History: Telling the Epics of Nature. BioScience doi: 10.1093/biosci/biu033 (full text, PDF)
Tschinkel, W.R. (2010) Back to basics: sociometry and sociogenesis of ant societies (Hymenoptera: Formicidae). Myrmecol. News 14:49-54, (full text, PDF)
Tschinkel, W.R. (2006). The Fire Ants, Belknap /Harvard University Press, 730 pp. (2007 Pulitzer Prize nominee; web page at Harvard University Press) The Fire Ants
Tschinkel, W.R. (1998) The reproductive biology of fire ant societies. Bioscience 48:593-605 (full text, PDF)
Tschinkel, W. R. (1991). Insect sociometry: a field in search of data. Insectes Soc. 38: 77-82 (full text, PDF)
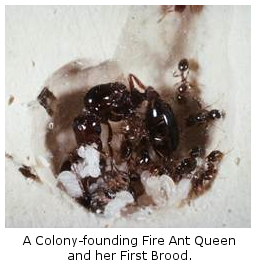
(2) Colony Founding: Years of experimentation and survey demonstrated unexpected complexity in the process of colony founding by newly-mated fire ant queens. Myriad interactions occur within founding colonies, including cannibalism of both queens and larvae. After the claustral period, brood raiding (brood stealing contests among newly-founded neighboring colonies) under natural conditions shapes the population dynamics of colony founding and early colony growth.
Tschinkel, W.R. (1998) An experimental study of pleometrotic colony-founding in the fire ant Solenopsis invicta: what is the basis for association?- Behav. Ecol. Sociobiol. 43:247-257 (full text, PDF)
Adams, E.S. and W.R. Tschinkel. (1995) Effects of foundress number on brood raids and queens survival in the fire ant Solenopsis invicta. Behav. Ecol. Sociobiol. 37:233-242 (full text, PDF)
Tschinkel, W. R. (1993) Resource allocation, brood production and cannibalism during colony founding in the fire ant, Solenopsis invicta. Behav. Ecol. Sociobiol. 33:209-223 (full text, PDF)
Tschinkel, W. R. (1992). Brood raiding and the population dynamics of founding and incipient colonies of the fire ant, Solenopsis invicta. Ecol. Entomol. 17: 179-188 (full text, PDF)
Porter, S. D. and W. R. Tschinkel. (1988). Adaptive value of nanitic workers in newly founded red imported fire ant colonies (Hymenoptera: Formicidae). Ann. Entomol. Soc. Amer. 79: 723-726 (full text, PDF)
Tschinkel, W. R. and D. F. Howard.(1983) Pleometrotic colony foundation in the fire ant, Solenopsis invicta. Behav. Ecol. Sociobiol., 12: 103-113. (full text, PDF)
 (3) A New Mode of Colony Founding in ants. The majority of
ants start new colonies by sending out newly mated queens, who start new
colonies from reserves stored in their bodies, without the aid of workers. In
addition to this well-known mode, fire ants also employ an alternate mode of founding
in which newly-mated queens seek out orphaned mature colonies. They enter
these and exploit the worker force to help them start their own colony, making
this a form of intraspecific social parasitism. Nothing like this has ever been
described before. It is likely that this phenomenon is widespread throughout
the ants, but has heretofore simply never been detected.
(3) A New Mode of Colony Founding in ants. The majority of
ants start new colonies by sending out newly mated queens, who start new
colonies from reserves stored in their bodies, without the aid of workers. In
addition to this well-known mode, fire ants also employ an alternate mode of founding
in which newly-mated queens seek out orphaned mature colonies. They enter
these and exploit the worker force to help them start their own colony, making
this a form of intraspecific social parasitism. Nothing like this has ever been
described before. It is likely that this phenomenon is widespread throughout
the ants, but has heretofore simply never been detected.
DeHeer, C.J. and W.R. Tschinkel. (1998) The success of alternative reproductive tactics in monogyne populations of the ant Solenopsis invicta: significance for transitions in social organization. Behav. Ecol. 9:130-135 (full text, PDF)
Tschinkel, W.R. (1996) A newly-discovered mode of colony founding among fire ants. Ins. Soc. 43:267-276 (full text, PDF)
McInnes, D.A. and W.R. Tschinkel. (1995)Queen dimorphism and reproductive strategies in the fire ant, Solenopsis geminata. Behav. Ecol. Sociobiol., 36:367-376 (full text, PDF)
 (4) Social Control
of Queen Fertility. There is only one individual in a fire ant colony that
lays eggs, and that is the queen. The rate at which she lays eggs must be
coordinated with the size of her work force. Through experiments, we
discovered and described the mechanism that coordinates the queen’s
egg-laying rate with the available brood-rearing labor, showing that the
metamorphosing larvae are the key to this regulation.
(4) Social Control
of Queen Fertility. There is only one individual in a fire ant colony that
lays eggs, and that is the queen. The rate at which she lays eggs must be
coordinated with the size of her work force. Through experiments, we
discovered and described the mechanism that coordinates the queen’s
egg-laying rate with the available brood-rearing labor, showing that the
metamorphosing larvae are the key to this regulation.
Tschinkel, W.R. (1995) Stimulation of fire ant queen fecundity by a highly specific brood stage. Ann. Entomol. Soc. Amer. 88:876-882 (full text, PDF)
Tschinkel, W. R. (1987).Social regulation of egg laying rate in queens of the fire ant, Solenopsis invicta. Physiol. Entomol. 13:237-250 (full text, PDF)
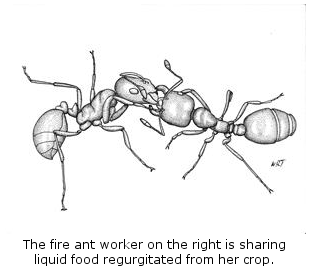 (5) Food Traffic. Among ants, only the oldest
workers leave the nest to forage. They bring liquid food back to the nest
in their crops and share it with nestmates through a complex web of
regurgitation. We illuminated the quantitative movement of food within the
colony, showed that the global patterns result from local decisions based on
local information, and discovered the “rules” by which food is shared with larvae
and other workers. This is also the first detailed study of the involvement of
larvae in the colony’s food web.
(5) Food Traffic. Among ants, only the oldest
workers leave the nest to forage. They bring liquid food back to the nest
in their crops and share it with nestmates through a complex web of
regurgitation. We illuminated the quantitative movement of food within the
colony, showed that the global patterns result from local decisions based on
local information, and discovered the “rules” by which food is shared with larvae
and other workers. This is also the first detailed study of the involvement of
larvae in the colony’s food web.
Cassill, D. L. and W. R. Tschinkel. (2000) Behavioral and developmental homeostasis in the fire ant, Solenopsis invicta. J. Insect Physiol. 46: 933-939 (full text, PDF)
Cassill, D.L. and W.R. Tschinkel. (1999) Food flow and information flow in colonies of the fire ant, Solenopsis invicta. Book chapter in Information Processing in Social Insects, Detrain and Deneubourg, eds., Birkhauser Verlag, Basel. pp. 69-81 (full text, PDF)
Cassill, D. L., and W. R. Tschinkel. (1995) Allocation of liquid food to larva via trophallaxis in the fire ant, Solenopsis invicta. Anim. Behav., 50:801-813 (full text, PDF)
Cassill, D. and W.R. Tschinkel. (1996) A duration constant for worker-to-larva trophallaxis in fire ants. Ins. Soc. 43:149-166 (full text, PDF)
Cassill, D.L. and W.R. Tschinkel. (1999). Task selection by workers of the fire ant, Solenopsis invicta. Behav. Ecol. Sociobiol. 45:301-310 (full text, PDF)
Cassill, D.L. and W.R. Tschinkel. (1999) Regulation of diet in the fire ant, Solenopsis invicta. J. Insect Behav. 12:307-328 (full text, PDF)
Cassill, D.L. and W.R. Tschinkel.(1999) Colony level effects on larval feeding in the fire ant, Solenopsis invicta. Insectes Sociaux. 46: 261-266. (full text, PDF)
Howard, D. F. and W. R. Tschinkel. (1981). Flow of food in colonies of the fire ant, Solenopsis invicta: A multi-factorial approach. Physiol. Entomol., 6: 297-306 (full text, PDF)
Howard, D. F. and W. R. Tschinkel. (1981). Internal storage of liquid food in isolated workers of the fire ant, Solenopsis invicta. J. Insect Physiol., 27: 67-74 (full text, PDF)
Glunn, F. J., D. F. Howard and W. R. Tschinkel. (1981). Food preference in colonies of the fire ant, Solenopsis invicta. Insectes Sociaux, 28: 217-222 (full text, PDF)
Howard, D. F. and W. R. Tschinkel. (1980).The effect of colony size and starvation on food flow in the fire ant, Solenopsis invicta. Behav. Ecol. Sociobiol., 7: 293-300 (full text, PDF)

(6) Evolution of Colony Life Histories: The methods of sociometry/sociogenesis, provide a simple procedure for the simultaneous determination of suites of colony characteristics and how they develop during colony growth. This is the social insect counterpart of embryonic development, and has received little attention. Sociometric/sociogenic studies on fire ants and harvester ants produced the first quantitative, full life-history descriptions of an ant colony, from birth to death.
Smith C.R. and W.R. Tschinkel, (2006). The sociometry and sociogenesis of reproduction in the Florida harvester ant (Pogonomyrmex badius) J. Insect Sci. 6:32 (full text, PDF)
Tschinkel, W.R. (1999).Sociometry and sociogenesis of colony-level attributes of the Florida harvester ant (Hymenoptera: Formicidae). Ann. Entom. Soc. Amer. 92:80-89 (full text, PDF)
Tschinkel, W.R. (1998). Sociometry and sociogenesis of colonies of the harvester ant, Pogonomyrmex badius:: worker characteristics in relation to colony size and season. Ins. Soc. 45:385-410 (full text, PDF)
Tschinkel, W. R. (1993).Sociometry and sociogenesis in colonies of the fire ant, Solenopsis invicta during one annual cycle.. Ecol. Monogr 63:425-457 (full text, PDF)
Tschinkel, W. R. (1991). Insect sociometry: a field in search of data. Insectes Soc. 38: 77-82 (full text, PDF)
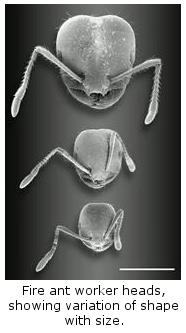 (7)
Worker Polymorphism. Workers in fire ant colonies vary
about 15-fold in weight from the smallest to the largest. This so-called
polymorphism develops gradually as colonies grow. Detailed descriptions
of polymorphism of colonies of a full range of sizes reveals how polymorphism
arises during colony development. Polymorphism has been hypothesized to
make colonies more efficient and therefore to increase their fitness. We tested
this in experiments that used brood rearing efficiency as a surrogate of
fitness.
(7)
Worker Polymorphism. Workers in fire ant colonies vary
about 15-fold in weight from the smallest to the largest. This so-called
polymorphism develops gradually as colonies grow. Detailed descriptions
of polymorphism of colonies of a full range of sizes reveals how polymorphism
arises during colony development. Polymorphism has been hypothesized to
make colonies more efficient and therefore to increase their fitness. We tested
this in experiments that used brood rearing efficiency as a surrogate of
fitness.
Tschinkel, W.R. (2013) The morphometry of Solenopsis fire ants. PlosOne 8(11): e79559. doi:10.1371/journal.pone.0079559. (full text, PDF)
Araujo, M. and W. R. Tschinkel (2010). Worker allometry in relation to colony size and social form in the fire ant, Solenopsis invicta, J. Insect Sci. 10:94 (full text, PDF)
Tschinkel W.R., A. Mikheyev and S. Storz. (2003) The allometry of worker polymorphism in the fire ant, Solenopsis invicta. J. Insect Sci. 3:2 (full text, PDF)
Tschinkel, W. R. (1988) Colony growth and the ontogeny of worker polymorphism in the fire ant, Solenopsis invicta Buren. Behav. Ecol. Sociobiol. 22: 103-115. (full text, PDF)
Porter, S. D. and W. R. Tschinkel. (1985).Fire ant polymorphism: the ergonomics of brood production. Behav. Ecol. Sociobiol. 16: 323-336. (full text, PDF)
Porter, S. D. and W. R. Tschinkel. (1985). Fire ant polymorphism: factors affecting worker size. Ann. Entomol. Soc. Amer. 78: 381-386. (full text, PDF)
Wood, L. A. and W. R. Tschinkel. (1981) Quantification and modification of worker size variation in the fire ant, Solenopsis invicta. Insectes Sociaux, 28: 117-128 (full text, PDF)
(8) Population Biology of Fire Ant Colonies. Ant populations consist of populations of colonies, as well as individual ants. There is thus an additional level of population processes in social insects, and these have rarely been studied. A five-year study of the population biology of fire ant colonies included the appearance, growth and decline of colonies, competitive territorial interactions, migration, the use of space and the relationship of territory size, colony size and neighborhoods.

A fire ant territory: boundaries shown by tape, colony by blue flag.
Adams, E.S. and W.R. Tschinkel. (2001). Mechanisms of population regulation in the fire ant Solenopsis invicta: an experimental study, J. Anim. Ecol. 70:355-369 (full text, PDF)
Adams, E. S., and W. R. Tschinkel. (1995). Density-dependent competition in fire ants: effects on colony survivorship and size variability. J. Anim. Ecol. 64:315-324 (full text, PDF)
Adams, E.S. and W.R. Tschinkel. (1995). Spatial dynamics of colony interactions in young populations of the fire ant, Solenopsis invicta. Oecologia, 102:156-163 (full text, PDF)
Tschinkel, W. R., E. S. Adams, and T. Macon. (1995). Territory area and colony size in the fire ant, Solenopsis invicta, J. Anim. Ecol., 64:473-480 (full text, PDF)
 (9)
Fire Ant, Native Ant Interactions. The fire ant is an
exotic invasive ant from Brazil that is of great political and economic
importance. Ecological work on this species has clearly demonstrated that
ecological disturbance favors the establishment and existence of this “weedy”
species, making humans the fire ant’s best friend. Moreover, the
long-held belief that fire ants suppress native ant populations was shown to be
false.
(9)
Fire Ant, Native Ant Interactions. The fire ant is an
exotic invasive ant from Brazil that is of great political and economic
importance. Ecological work on this species has clearly demonstrated that
ecological disturbance favors the establishment and existence of this “weedy”
species, making humans the fire ant’s best friend. Moreover, the
long-held belief that fire ants suppress native ant populations was shown to be
false.
King, J.R. and Tschinkel, W.R. (2016) Experimental evidence that dispersal drives ant community assembly in human- altered ecosystems. Ecology 97: 236-249. (full text, PDF)
Tschinkel W.R. and King J.R. (2015). Ant community and habitat limit colony establishment by the fire ant, Solenopis invicta. Functional Ecology, doi:10.1111/1365-2435.12794. (full text, PDF)
King, J.R. and W. R. Tschinkel (2013) Experimental evidence that human impacts drive fire ant invasions and ecological change: Response. J. Anim. Ecol., in press. (full text, PDF)
Tschinkel, W.R. and King, J.R. (2013) The role of habitat in the persistence of fire ant (Solenopsis invicta) populations. PlosOne 8(10): e78580. doi:10.1371/journal.pone.0078580. (full text, PDF)
King, J.R. and W. R. Tschinkel (2013) Experimental evidence for weak effects of fire ants in a naturally invaded pine-savanna ecosystem in north Florida. Ecol. Entomol. 38: 68–75 (full text, PDF)
King, J.R., W. R. Tschinkel and K.G. Ross (2009) A case study of human exacerbation of the invasive species problem: transport and establishment of polygyne fire ants in Tallahassee, Florida, USA. Biol. Invasions 11:373-377. (full text, PDF)
King, J.R. and W. R. Tschinkel (2008). Experimental evidence that human impacts drive fire ant invasions and ecological change. PNAS 105:20339-20343. (full text, PDF)
Tschinkel, W.R. and J.R. King (2007). Targeted removal of ant colonies in ecological experiments, using hot water. Journal of Insect Science, online at http://jinsectscience.oxfordjournals.org/content/7/1/41/ (full text, PDF)
King, J.R. and W.R. Tschinkel, (2006). Experimental evidence that the introduced fire ant, Solenopsis invicta, does not competitively suppress co-occurring ants in a disturbed habitat, J. Animal Ecol. 75: 1370–1378 (full text, PDF)
Lubertazzi, D. and W.R. Tschinkel. (2003).Ant community change across a ground vegetation gradient in north Florida's longleaf pine flatwoods. J. Insect Science 3:21 (full text, PDF)
Tschinkel, W.R. (1998) The reproductive biology of fire ant societies. Bioscience 48:593-605 (full text, PDF)
Tschinkel, W. R. (1993).The fire ant: still unvanquished. Proc. Indiana Acad. Sci., in Biological Pollution: The Control and Impact of Invasive Exotic Species, ed. B.N. McKnight, Indiana Acad. Sci., Indianapolis. (full text, PDF)
Tschinkel, W. R. (1988).Distribution of the fire ants Solenopsis invicta and S. geminata in north Florida in relation to habitat and disturbance. Ann. Entomol. Soc. Amer. 81: 76-81. (full text, PDF)
Tschinkel, W. R. (1986) The ecological nature of the fire ant, some aspects of colony function and some unanswered questions, in Fire Ants and Leaf-cutter Ants: Biology and Management, C. S. Lofgren and R. K. Vander Meer, eds. Westview Press, 1986.
 (10)
Other Fire ant Biology. In spite of the
importance of fire ants, much of their biology is unknown or poorly
understood. A number of our papers illuminate important aspects of the
biology of this ant, including its capacity to regulate larval development by
choosing developmental temperatures, the longevity of its queens and colonies,
its sanitation behavior, its brood-care signals, sperm storage, food storage,
organization of foraging and the effect of temperature on foraging.
(10)
Other Fire ant Biology. In spite of the
importance of fire ants, much of their biology is unknown or poorly
understood. A number of our papers illuminate important aspects of the
biology of this ant, including its capacity to regulate larval development by
choosing developmental temperatures, the longevity of its queens and colonies,
its sanitation behavior, its brood-care signals, sperm storage, food storage,
organization of foraging and the effect of temperature on foraging.
Tschinkel WR (2018) Testing the effect of a nest architectural feature in the fire ant Solenopsis invicta (Hymenoptera:Formicidae). Myrmecol. News 27: 1-5(full text, PDF)
Tschinkel, W.R. (2010) The organization of foraging in the fire ant, Solenopsis invicta. J. Insect Sci. 10:26 online at http://jinsectscience.oxfordjournals.org/content/11/1/26 (full text, PDF)
Gayahan, G.G, and W.R. Tschinkel (2008) Fire ants (Solenopsis invicta) dry and store insect pieces for later use, J. Insect Sci. online at http://jinsectscience.oxfordjournals.org/content/8/1/39 (full text, PDF)
Penick, C.A. and W. R. Tschinkel (2008) Thermoregulatory brood transport in the fire ant, Solenopsis invicta, Insectes Sociaux 55:175-172. (full text, PDF)
Tschinkel, W.R. and J.R. King (2007). Targeted removal of ant colonies in ecological experiments, using hot water. Journal of Insect Science, online at http://jinsectscience.oxfordjournals.org/content/7/1/41 (full text, PDF)
Haight, K.L. and W. R. Tschinkel. (2003) Patterns of venom synthesis and use in the fire ant, Solenopsis invicta. Toxicon 42:673-683 (full text, PDF)
McInnes, D.A. and W.R. Tschinkel. (1995). Queen dimorphism and reproductive strategies in the fire ant, Solenopsis geminata. Behav. Ecol. Sociobiol., 36:367-376 (full text, PDF)
Porter, S. D., and W. R. Tschinkel. (1993).Fire ant thermal preferences: behavioral control of growth and metabolism. Behav. Ecol. Sociobiol. 32:321-329. (full text, PDF)
Tschinkel, W. R. (1993). The fire ant: still unvanquished. Proc. Indiana Acad. Sci., in Biological Pollution: The Control and Impact of Invasive Exotic Species, ed. B.N. McKnight, Indiana Acad. Sci., Indianapolis (full text, PDF)
Tschinkel, W. R., and S. D. Porter. (1988). The efficiency of sperm use in queens of the fire ant, Solenopsis invicta Buren. Ann. Entomol. Soc. Amer. 81: 777-781. (full text, PDF)
Porter, S. D., and W. R. Tschinkel. (1987).Foraging in Solenopsis invicta (Hymenoptera: Formicidae): effects of weather and season. Envir. Entomol. 16: 802-808. (full text, PDF)
Tschinkel, W. R. (1987). Fire ant queen longevity and age: estimation by sperm depletion. Ann. Entomol. Soc. Amer. 80: 263-266. (full text, PDF)
Tschinkel, W. R. and D. R. Howard. (1980) .A simple, non-toxic home remedy against fire ants. J. Ga. Entomol. Soc. 15:102-105 (full text, PDF)
Howard, D. R. and W. R. Tschinkel. (1976).Aspects of necrophoric behavior in the red imported fire ant, Solenopsis invicta. Behaviour 56: 157-180. (full text, PDF)
Walsh, J. P. and W. R. Tschinkel. (1974). Brood recognition by contact pheromone in the imported fire ant, Solenopsis invicta. Animal Behaviour 22: 695-704. (full text, PDF)
 (11)
Architecture of Subterranean Ant Nests. We have
pioneered the study of nest architecture of ground-nesting ants through the
creation of nest casts using orthodontal plaster or liquid metal. This
work is still in progress, but has received national and international
attention. The initial goal of this research is to provide an inventory of the
structures ants create, as well as an understanding of how colonies are organized
to produce these structures.
(11)
Architecture of Subterranean Ant Nests. We have
pioneered the study of nest architecture of ground-nesting ants through the
creation of nest casts using orthodontal plaster or liquid metal. This
work is still in progress, but has received national and international
attention. The initial goal of this research is to provide an inventory of the
structures ants create, as well as an understanding of how colonies are organized
to produce these structures.
Tschinkel WR (2017) Do Florida harvester ant colonies (Pogonomyrmex badius) have a nest architecture plan? Ecology 98: 1176–1178. (full text, PDF)
Tschinkel WR (2015) Biomantling and bioturbation by colonies of the Florida harvester ant, Pogonomyrmex badius. PLoS One 10(3): e0120407. doi:10.1371/journal.pone.0120407 (full text, PDF)
Tschinkel WR (2014) Nest relocation and excavation in the Florida harvester ant, Pogonomyrmex badius. PLoS One 9(11): e112981. doi:10.1371/journal (full text, PDF)
Tschinkel, W.R. 2013. Florida harvester ant nest architecture, nest relocation and soil carbon dioxide gradients. Online at PlosOne 8(3): e59911. doi:10.1371/journal.pone.0059911. (full text, PDF)
Tschinkel W. R., Rink W. J., Kwapich C. L. (2015) Subterranean transport of sand and seeds by caching in the harvester ant, Pogonomyrmex badius, PlosOne (in press, July 2015). (full text, PDF)
Tschinkel, W.R. (2015) The architecture of subterranean ant nests: beauty and mystery underfoot. Journal of Bioeconomics. In press. (full text, PDF)
Tschinkel WR (2015) Biomantling and bioturbation by colonies of the Florida harvester ant, Pogonomyrmex badius. PLoS One 10(3): e0120407. doi:10.1371/journal.pone.0120407 (full text, PDF)
Tschinkel WR (2014) Nest relocation and excavation in the Florida harvester ant, Pogonomyrmex badius. PLoS One 9(11): e112981. doi:10.1371/journal. (full text, PDF)
Tschinkel, W.R. (2013). Florida harvester ant nest architecture, nest relocation and soil carbon dioxide gradients. Online at PlosOne 8(3): e59911. doi:10.1371/journal.pone.0059911. or http://www.plosone.org/article/info%3Adoi%2F10.1371%2Fjournal.pone.0059911 (full text, PDF)
Tschinkel, W.R. (2013) A method for using ice to construct subterranean ant nests (Hymenoptera: Formicidae) and other soil cavities. Myrmecol. News 18: 99-102. (full text, PDF)
Rink W. J, Dunbar, J.S., Tschinkel W.R., Kwapich C., Repp A., Stanton W. and Thulman, D.K. (2013) Subterranean transport and deposition of quartz by ants in sandy sites relevant to age overestimation in optical luminescence dating. J. Archeol. Sci. 40: 2217–2226 (full text, PDF)
Tschinkel, W.R. (2011) The nest architecture of three species of Aphaenogaster in north Florida. J. Insect Sci. online: http://jinsectscience.oxfordjournals.org/content/11/1/105 (full text, PDF)
Tschinkel, W. R. (2010) Methods for casting subterranean ant nests. J. Insect Sci. 10:88 online at: http://jinsectscience.oxfordjournals.org/content/10/1/88 (full text, PDF)
Cerquera, L.M. and W.R. Tschinkel (2009). The nest architecture of Odontomachus brunneus. J. Insect Sci. 10:64. online at http://jinsectscience.oxfordjournals.org/content/10/1/64 (full text, PDF)
Tschinkel, W.R. (2005) The Nest Architecture of the Ant, Camponotus socius. J. Insect Science 5:9 (full text, PDF)
Tschinkel, W.R. (2004). The Nest Architecture of the Florida Harvester Ant, Pogonomyrmex badius. J. Insect Science 4:21 (full text, PDF)
Mikheyev, A. and W. R. Tschinkel. (2004). Nest architecture of the ant Formica pallidefulva: structure, costs and rules of excavation. Insectes Sociaux 51:30-36 (full text, PDF)
Tschinkel, W.R. (2003) Subterranean Ant Nest Architecture: Trace Fossils of the Future? Paleogeogr. Paleoclim. Paleoecol. 192:321-333 (full text, PDF) not working
Cassill, D.L., W.R. Tschinkel and S.B. Vinson. (2002)Nest complexity, group size and brood rearing in the fire ant, Solenopsis invicta. Insectes Soc. 49(2):158-163 (full text, PDF)
Tschinkel, W.R. (2001) Colonies in space. Nat. Hist.
Tschinkel, W.R. (1998).Sociometry and sociogenesis of colonies of the harvester ant, Pogonomyrmex badius, in relation to colony size and season: distribution of workers and seeds in the nest. Insectes Sociaux. 45:385-410 (full text, PDF)
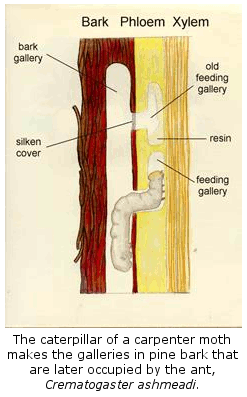 (12) Natural
History of Arboreal Ants. With the discovery by Dr.
Frances James and Charles Hess that the endangered red-cockaded woodpecker ate
mostly a single species of tree-dwelling ant, Crematogaster ashmeadi, it
became important to learn as much as possible about the ecology and natural history
of this critical ant. Subsequently, we have worked out most of the basic
outlines of the colony cycle, natural history, distribution and abundance of
this ant. In the process, we discovered that this previously unknown ant
is the most abundant and dominant arboreal ant in the coastal plain pine
forests of the southern states.
(12) Natural
History of Arboreal Ants. With the discovery by Dr.
Frances James and Charles Hess that the endangered red-cockaded woodpecker ate
mostly a single species of tree-dwelling ant, Crematogaster ashmeadi, it
became important to learn as much as possible about the ecology and natural history
of this critical ant. Subsequently, we have worked out most of the basic
outlines of the colony cycle, natural history, distribution and abundance of
this ant. In the process, we discovered that this previously unknown ant
is the most abundant and dominant arboreal ant in the coastal plain pine
forests of the southern states.
Tschinkel, W.R. (2002). The Natural History of the Arboreal Ant, Crematogaster ashmeadi (Hymenoptera: Formicidae). J. Insect Sci. 2:12 (full text, PDF)
Baldacci, J. and W.R. Tschinkel. (1999).An experimental study of colony founding in pine saplings by queens of the arboreal ant, Crematogaster ashmeadi. Ins. Soc. 46:41-44 (full text, PDF)
Tschinkel, W.R. and C. Hess. (1999)The arboreal ant community of a north Florida pine forest, with emphasis on Crematogaster ashmeadi. Ann. Entomol. Soc. Amer. 92:63-70 (full text, PDF)
Hahn, D.A. and W.R. Tschinkel. (1997) Settlement and distribution of newly-mated queens of the arboreal ant, Crematogaster ashmeadi, in a longleaf pine forest. Ins. Soc. 44:323-336 (full text, PDF)
(13) Natural History
of Other Ants. The natural history of most ant species is, at best, poorly
known. Adding to the store of natural history knowledge allows inferences
about the evolution of ant sociality, ecology and behavior to be drawn. A
number of “background” projects on the natural history of local species  of ants have
revealed novel and fascinating things about the lives of Florida’s ants.
of ants have
revealed novel and fascinating things about the lives of Florida’s ants.
Mason, K. S., Kwapich, C. L., Tschinkel, W.R. (2015) Respiration, worker body size, tempo and activity in whole colonies of ants. Physiol. Entomol. 40: 149–165, DOI: 10.1111/phen.12099. (full text, PDF)
Murdock, T. and W.R. Tschinkel (2015). Seasonal life history and nest architecture of the ant, Pheidole morrisi. Insectes Sociaux, DOI 10.1007/s00040-015-0403-9. (full text, PDF)
Tschinkel, W.R., T. Murdock, J.R. King and C. Kwapich (2012). Ants and groundwater in the north Florida flatwoods. Journal of Insect Science12:114. (full text, PDF)
Hart,L. M. and W. R. Tschinkel (2012) A seasonal natural history of the ant, Odontomachus brunneus. Insectes Sociaux. 59:45–54 (full text, PDF)
Laskis, K.O. and W. R. Tschinkel (2009). The Seasonal Natural History of the Ant, Dolichoderus mariae (Hymenoptera: Formicidae) In Northern Florida, J. Insect Sci. (full text, PDF)
King, J.R. and W.R. Tschinkel (2007). Range expansion and local population increase of the exotic ant, Pheidole obscurithorax, in the southeastern United States. Florida Entomologist 90: 435-439. (full text, PDF)
Storz, S. and W.R. Tschinkel. (2003) Distribution, spread, and ecological associations of the introduced ant Pheidole obscurithorax in the southeastern United States. J. Insect Sci. 4:12 (full text, PDF)
Tschinkel, W.R. (2002).The Natural History of the Arboreal Ant, Crematogaster ashmeadi (Hymenoptera: Formicidae). J. Insect Sci. 2:12 (full text, PDF)
Powell, S. and W. R. Tschinkel. (1999) Ritualized conflict in Odontomachus brunneus and the generation of interaction-based task allocation: a new organizational mechanism in ants. Anim. Behaviour 58: 965–972. (full text, PDF)
McInnes, D.A. and W.R. Tschinkel. (1996) Mermithid nematode parasitism of Solenopsis ants (Hymenoptera: Formicidae) of northern Florida. Ann. Entomol. Soc. Amer. 89:231-237 (full text, PDF)
McInnes, D.A. and W.R. Tschinkel. (1995) Queen dimorphism and reproductive strategies in the fire ant, Solenopsis geminata. Behav. Ecol. Sociobiol., 36:367-376 (full text, PDF)
Hood, W. G., and W. R. Tschinkel. (1990). Desiccation resistance in arboreal and terrestral ants. Physiol. Entomol. 15: 23-35 (full text, PDF)
Tschinkel, W. R. (1987). Seasonal life history and nest architecture of a cold-loving ant, Prenolepis imparis. Insectes Soc. 34: 143-164 (full text, PDF)
Tschinkel, W. R. (1987). The relationship between ovariole number and spermathecal sperm count in ant queens: a new allometry. Ann. Entomol. Soc. Amer. 80: 208-211 (full text, PDF)
Tschinkel, W. R. and A. Bhatkar. (1974). Oriented mound-building in the ant, Trachymyrmex septentrionalis. Environ. Entomol. 3: 667-673 (full text, PDF)
 (14)
Fungus gardening ants. We have studied the natural
history of some local ants more intensively than others. Two in
particular stand out--- the Florida harvester ant (see below), and the fungus
gardening ant, Trachymyrmex septentrionalis. The fungus-gardening
ant collects plant bits and caterpillar droppings for its fungus gardens,
raising the question, “does it know what is good for its fungus?” Because these
ant nests are so numerous, they are a major agent of biomantling and
bioturbation.
(14)
Fungus gardening ants. We have studied the natural
history of some local ants more intensively than others. Two in
particular stand out--- the Florida harvester ant (see below), and the fungus
gardening ant, Trachymyrmex septentrionalis. The fungus-gardening
ant collects plant bits and caterpillar droppings for its fungus gardens,
raising the question, “does it know what is good for its fungus?” Because these
ant nests are so numerous, they are a major agent of biomantling and
bioturbation.
Tschinkel, W.R. and Seal, J.N. (2016) Bioturbation by the fungus-gardening ant, Trachymyrmex septentrionalis. PLoS ONE 11(7): e0158920. doi:10.1371/journal.pone.0158920.(full text, PDF)
Seal, J.N. and W.R. Tschinkel (2010) Distribution of the fungus-gardening ant (Trachymyrmex septentrionalis) during and after a record drought. Insect Conservation and Diversity (2010) 3:134-142. (full text, PDF)
Seal, J.N. and W. R. Tschinkel (2008). Food limitation in the fungus gardening ant, Trachymyrmex septentrionalis. Ecol. Entomol. 33:597-607. (full text, PDF)
Seal, J.N. and W.R. Tschinkel, (2007) Energetics of newly mated queens and colony founding in the fungus-gardening ants Cyphomyrmex rimosus and Trachymyrmex septentrionalis (Hymenoptera: Formicidae). Physiological Entomology, 32: 8-15. (full text, PDF)
Seal, J.N. and W.R. Tschinkel (2007) Complexity in an obligate mutualism: Do fungus-gardening ants know what makes their garden grow? Behav. Ecol. Sociobiol. 61:1151–1160. (full text, PDF)
Seal, J.N. & Tschinkel, W.R. (2007) Co-evolution and the superorganism: switching cultivars does not alter the performance of fungus-gardening ant colonies. Functional Ecology, 21, 988-997. (full text, PDF)
Seal J.N. and Tschinkel W.R. (2006) Energetics of newly mated queens and colony founding in the fungus-gardening ants Cyphomyrmex rimosus and Trachymyrmex septentrionalis (Hymenoptera: Formicidae). Physiol Entomol. 32: 8-15. (full text, PDF)
Seal, J.N. and W.R. Tschinkel, (2006). Complexity in an obligate mutualism: Do fungus-gardening ants know what makes their garden grow? Behav. Ecol. Sociobiol. 61:1151–1160. (full text, PDF)
Seal J.N. and Tschinkel W.R. (2006) Colony productivity of the fungus-gardening ant, Trachymyrmex septentrionalis McCook, in a Florida pine forest (Hymenoptera: Formicidae). Ann Entomol Soc Am: 99: 673-682 (full text, PDF)
Tschinkel, W. R. and A. Bhatkar. Oriented mound‑building in the ant, Trachymyrmex septentrionalis. Environ. Entomol. 3: 667‑673 (1974). (full text, PDF)
 (15)
Harvester ant biology. The Florida harvester
ant, Pogonomymex badius is a large, charismatic ant of the sandhills
ecosystem of the Gulf Coastal Plain. It collects seeds and stores them in
underground chambers for future use, excavates a large, beautiful subterranean
nest, decorates its nest disc with bits of charcoal and attracts many inquilines
and myrmecophiles that live in its nest.
(15)
Harvester ant biology. The Florida harvester
ant, Pogonomymex badius is a large, charismatic ant of the sandhills
ecosystem of the Gulf Coastal Plain. It collects seeds and stores them in
underground chambers for future use, excavates a large, beautiful subterranean
nest, decorates its nest disc with bits of charcoal and attracts many inquilines
and myrmecophiles that live in its nest.
Kwapich C.L. and Tschinkel W.R. (2016) Limited flexibility and unusual longevity shape forager allocation in the Florida harvester ant (Pogonomyrmex badius) Behavioral Ecology and Sociobiology 70: 221-225. DOI 10.1007/s00265-015-2039-1.(full text, PDF)
Tschinkel, W.R. and Kwapich, C.L. (2016) The Florida harvester ant, Pogonomyrmex badius, relies on germination to consume large seeds. PloSOne 11(11): e0166907. doi:10.1371/journal.pone.0166907. (full text, PDF)
Tschinkel W.R. and Dominguez, D.J. (2017) An illustrated guide to seeds found in nests of the Florida harvester ant, Pogonomyrmex badius. PloS one 12.3 (2017): e0171419.(full text, PDF)
Tschinkel WR (2017) Life span, age, size-specific mortality and dispersion of colonies of the Florida harvester ant, Pogonomyrmex badius. Insectes Sociaux, 64: 285–296.(full text, PDF)
Tschinkel WR, Hanley N (2017) Vertical organization of the division of labor within nests of the Florida harvester ant, Pogonomyrmex badius. PLoS ONE 12(11): e0188630. https://doi.org/10.1371/journal.pone.0188630 (full text, PDF)
Tschinkel W. R., Rink W. J., Kwapich C. L. (2015) Subterranean transport of sand and seeds by caching in the harvester ant, Pogonomyrmex badius, PlosOne (in press, July 2015) (full text, PDF)
Tschinkel WR (2015) Biomantling and bioturbation by colonies of the Florida harvester ant, Pogonomyrmex badius. PLoS One 10(3): e0120407. doi:10.1371/journal.pone.0120407 (full text, PDF)
Tschinkel WR (2014) Nest relocation and excavation in the Florida harvester ant, Pogonomyrmex badius. PLoS One 9(11): e112981. doi:10.1371/journal (full text, PDF)
Tschinkel, W.R. 2013. Florida harvester ant nest architecture, nest relocation and soil carbon dioxide gradients. Online at PlosOne 8(3): e59911. doi:10.1371/journal.pone.0059911. (full text, PDF)
Rink W. J, Dunbar, J.S., Tschinkel W.R., Kwapich C., Repp A., Stanton W. and Thulman, D.K. (2013) Subterranean transport and deposition of quartz by ants in sandy sites relevant to age overestimation in optical luminescence dating. J. Archeol. Sci. 40: 2217–2226 (full text, PDF)
Kwapich, C.M. and W. R. Tschinkel (2013) Demography, demand, death and the seasonal allocation of labor in the Florida harvester ant (Pogonomyrmex badius). Behav. Ecol. Sociobiol. 67:2011-2027. DOI: 10.1007/s00265-013-1611-9. (full text, PDF)
Smith C. R. and W. R. Tschinkel, (2007). The adaptive nature of non-food collection for the Florida harvester ant, Pogonomyrmex badius. Ecological Entomology 32:105-112 (full text, PDF)
Smith C.R. and W.R. Tschinkel, (2006) The sociometry and sociogenesis of reproduction in the Florida harvester ant (Pogonomyrmex badius) J. Insect Sci. 6:32. (full text, PDF)
Smith C. R. and W. R. Tschinkel, (2005) Object depots in the genus Pogonomyrmex: Exploring the “who”, what, when, and where. J. Insect Behav. 18:859-879 (full text, PDF)
Tschinkel, W.R. The Nest Architecture of the Florida Harvester Ant, Pogonomyrmex badius. J. Insect Science 4:21 (2004). http://jinsectscience.oxfordjournals.org/content/4/1/21 (full text, PDF)
Tschinkel, W.R. (1999) Sociometry and sociogenesis of colony-level attributes of the Florida harvester ant (Hymenoptera: Formicidae). Ann. Entom. Soc. Amer. 92:80-89. (full text, PDF)
Tschinkel, W.R. (1998) Sociometry and sociogenesis of colonies of the harvester ant, Pogonomyrmex badius: worker characteristics in relation to colony size and season. Ins. Soc. 45:385-410. (full text, PDF)
Tschinkel, W.R. (1998) Sociometry and sociogenesis of colonies of the harvester ant, Pogonomyrmex badius, in relation to colony size and season: distribution of workers and seeds in the nest. Ecol. Entomol. 24: 222-237. (full text, PDF)
 (16)
Chemical Defense, Pheromones and Systematics
of Tenebrionid and Other Beetles. Tenebrionid beetles are a cosmopolitan
family of about 20,000 species, most of whom possess a chemical defensive
system. We carried out the largest and most complete comparative study of this
chemical defense system, including analysis of the chemical composition of the
secretion, anatomy of the glands, and defensive behavior. This remains a
standard-setting study that has rarely been equaled or surpassed. These
comparative studies culminated in the revision of the higher systematics of
family Tenebrionidae.
(16)
Chemical Defense, Pheromones and Systematics
of Tenebrionid and Other Beetles. Tenebrionid beetles are a cosmopolitan
family of about 20,000 species, most of whom possess a chemical defensive
system. We carried out the largest and most complete comparative study of this
chemical defense system, including analysis of the chemical composition of the
secretion, anatomy of the glands, and defensive behavior. This remains a
standard-setting study that has rarely been equaled or surpassed. These
comparative studies culminated in the revision of the higher systematics of
family Tenebrionidae.
Hill, C. S. and W. R. Tschinkel. (1985). Defensive secretion production in the tenebrionid beetle, Zophobas atratus: effects of age, sex and milking frequency. J. Chem. Ecol. 11: 1083-1092 (full text, PDF)
Doyen, J. T. and W. R. Tschinkel. (1982). Phenetic and cladistic relationships among tenebrionid beetles (Coleoptera). System. Entomol. 7: 127-183 (full text, PDF)
Blum, M. S., T. H. Jones, G. J. House and W. R. Tschinkel. (1981) Defensive secretions of tiger beetles: cyanogenetic basis. Comp. Biochem. Physiol., 69B: 903-904 (full text, PDF)
Tschinkel, W. R. and J. T. Doyen. (1980).Comparative anatomy of the defensive gland, ovipositor and female genital tube of the tenebrionid beetles. Intern. J. Insect Morphol. Embryol. 9: 321-369 (full text, PDF)
Lloyd, H S., S. L. Evans, A. H. Khan, W. R. Tschinkel and M. S. Blum. (1978). 8-Hydroxyisocoumarin and 3,4-dihydro-8-hydroxyisocoumarin in the defensive secretion of the tenebrionid beetle, Apsena pubescens. Insect Biochem. 8: 333-336 (full text, PDF)
Eisner, T., T. H. Jones, D. J. Aneshansley, W. R. Tschinkel, R. E. Silberglied and J. Meinwald. (1977). Chemistry of defensive secretions of bombardier beetles (Brachinini, Metriini, Ozaenini, Paussini). J. Insect Physiol. 23: 1383-1386 (full text, PDF)
Tschinkel, W. R. (1975).A comparative study of the chemical defensive system of tenebrionid beetles. I. Chemistry of the secretion. J. Insect Physiol. 21: 753-783 (full text, PDF)
Tschinkel, W. R. (1975).A comparative study of the chemical defensive system of tenebrionid beetles. III. Morphology of the glands. J. Morphol. 145: 355-370 (full text, PDF)
Tschinkel, W. R. (1975).A comparative study of the chemical defensive system of tenebironid beetles. II. Defensive behavior and ancillary features. Ann. Entomol. Soc. Amer. 68: 439-453 (full text, PDF)
Tschinkel, W. R. (1975) Unusual occurrence of aldehydes and ketones in the defensive secretion of the tenebrionid beetle, Eleodes beameri. J. Insect Physiol. 21: 659-671 (full text, PDF)
Tschinkel, W. R. (1972) .6-Alkylated-naphthoquinones from defensive secretion of the tenebrionid beetle, Argoporis alutacea. J. Insect Physiol. 18: 711-722 (full text, PDF)
Tschinkel, W. R. (1970) Chemical studies on the sex pheromone of Tenebrio molitor (Coleoptera: Tenebrionidae). Ann. Ent. Soc. Amer. 63: 626-627 (full text, PDF)
Tschinkel, W. R. (1969) Phenols and quinones from the defensive secretion of the tenebrionid beetle, Zophobas rugipes. J. Insect Physiol. 15: 191-200 (full text, PDF)
Tschinkel, W. R., C. D. Willson and H. A. Bern. (1967) Sex pheromone of the mealworm beetle (Tenebrio molitor). J. Exp. Zool. 164: 81-86 (full text, PDF)
 (17)
Inhibition of Metamorphosis by Crowding.
Crowding in tenebrionid beetle larvae has a novel effect in that it directly
inhibits metamorphosis through the effects of larval-larval contacts. We worked
out its natural history, and through experimentation, described the mechanism
of the inhibition and its life-history consequences in Zophobas atratus.
WE also demonstrated the importance of this effect in natural populations.
(17)
Inhibition of Metamorphosis by Crowding.
Crowding in tenebrionid beetle larvae has a novel effect in that it directly
inhibits metamorphosis through the effects of larval-larval contacts. We worked
out its natural history, and through experimentation, described the mechanism
of the inhibition and its life-history consequences in Zophobas atratus.
WE also demonstrated the importance of this effect in natural populations.
Tschinkel, W. R. (1993) Crowding, maternal age and age at pupation in the life history of Zophobas rugipes (Coleoptera: Tenebironidae). Ann. Entomol. Soc. Amer. 86:278-297 (full text, PDF)
Tschinkel, W. R. (1981).Larval dispersal and cannibalism in natural population of Zophobas atratus (Coleoptera: Tenebrionidae). Anim. Behavior 29:990-996 (full text, PDF)
Tschinkel, W. R. (1978). Dispersal behavior of the larval tenebrionid beetle, Zophobas rugipes. Physiol. Zool. 51: 300-313 (full text, PDF)
Tschinkel, W. R. and G. van Belle. (1976).Dispersal of larvae of the tenebrionid beetle, Zophobas rugipes, in relation to weight and crowding. Ecology 57: 161-168 (full text, PDF)
Tschinkel, W. R. and C. D. Willson. (1971). Inhibition of pupation due to crowding in some tenebrionid beetles. J. Exp. Zool. 176: 137-146 (full text, PDF)
(18) Causes and characteristics of Namibian fairy circles. The Namib Desert of The eastern margin of the Namib Desert of Namibia, Africa is home to a mysterious phenomenon in which the grassy vegetation is punctuated by tens of thousands of evenly spaced barren spots surrounded by taller grass. The origin of these circles is unknown, although many hypotheses have been offered. Using satellite images and visits on the ground, I am describing the range of variation of fairy circles and estimating their life span.

Fairy circles in the Namib Desert of Namibia.
Tschinkel W.R. (2015) Experiments testing the causes of Namibian fairy circles. PloS One 10 (10):e0140099. http://journals.plos.org/plosone/article?id=10.1371/journal.pone.0140099 (full text, PDF)
Cramer, M. D., Barger, N. N. and Tschinkel, W. R. (2016), Edaphic properties enable facilitative and competitive interactions resulting in fairy circle formation. Ecography. doi:10.1111/ecog.02461.(full text, PDF)
Tschinkel W.R. (2015) Experiments testing the causes of Namibian fairy circles. PloS One (full text, PDF)
Tschinkel, W.R. (2012) The life cycle and life span of Namibian fairy circles. PLoS ONE 7(6): e38056. doi:10.1371/journal.pone.0038056 . Link
Tschinkel, W. R. (2010) The foraging tunnel system of the Namibian termite, Baucaliotermes hainesi. J. Insect Sci. 10:65. online at: http://jinsectscience.oxfordjournals.org/content/10/1/65 (full text, PDF)
(19) Odds and Ends: Occasional opportunistic research into unusual areas that are not the normal focus of our research.
Hess C.A. and Tschinkel W.R. (2016) Effect of thinning and clear-cuts on the transmission of fire in slash pine plantations during restoration to longleaf pine. Ecological Rest. 2017: 33-40. (full text, PDF)
Nierenberg, N, W. R. Tschinkel and V. J. Tschinkel (2010). Early Climate Change Consensus at the National Academy: The Origins and Making of Changing Climate, Historical Studies in the Natural Sciences, 40 (3):318–349 (full text, PDF)
Tschinkel, W. R. (1984) .Zophobas rugipes and Z. atratus are the same species. Coleop. Bull. 38: 325-333 (full text, PDF)
Tschinkel, W. R. (1978). Ovoviviparity in some tenebrionid beetles. Coleopt. Bull. 32: 315-318 (full text, PDF)
Tschinkel, W. R. and J. T. Doyen. (1976). Sound production by substratal tapping in the tentyriid beetles of the genus Eusattus. Coleopt. Bull. 30: 331-335 (full text, PDF)
Doyen, J. T. and W. R. Tschinkel. (1974). Population size, microgeographic distribution and habitat separation in some tenebrionid beetles (Coleoptera). Ann. Entomol. Soc. Amer. 67: 617-626 (full text, PDF)
Tschinkel, W. R. and P. G. Close. (1973). The trail pheromone of the termite, Trinervitermes trinervoides. J. Insect Physiol. 19: 707-721 (full text, PDF)
Tschinkel, W. R. (1973). The sorption of water vapor by windborne plant debris in the Namib Desert. Madoqua II 2: 21-24 (full text, PDF)
The Fire Ants (Harvard/Belknap Press, 2006). This large volume brings together and interprets all current knowledge of fire ant biology for both the professional biologist and the educated lay reader. It was nominated for a Pulitzer Prize in September 2006.

Dust jacket of The Fire Ants.
From Reviews of The Fire Ants
- In his foreword to the book, E.O. Wilson wrote "Tschinkel has delivered a masterpiece" and went on to say that "this is how future biology will be written: a holistic account of a species or group of species across all levels of biological organization…”
- James Gorman, NY Times science writer, in an extremely positive review, said, "This is what the public needs to know about science, not just the results presented in the driest form possible."
- The Times Literary Supplement (London) wrote: "This book is a masterly and detailed account of some of nature's greatest opportunists, the fire ants." Referring to the anecdotal and less-technical "interludes" sprinkled throughout the book, the TLS called them "highly readable primers… that give the flavour of the man beautifully", and then went on to write, "No doubt, the stuffed shirt faction of academic entomologists will harrumph and snort at the sometimes folksy style, but that is their problem.”
- “This is a wonderful book, comprehensive in its coverage of fire ant social biology, extraordinarily lucid in its description of complex topics, and beautifully synthetic in tying together the disparate threads of evidence relevant to the discussion of each topic.”
- “This book is without parallel as a thorough description of the biology of an important social insect.”
- “Not only is the author, Walter Tschinkel, an expert on Fire Ants, he is a skillful writer. His ability to sprinkle humor in every chapter makes the book enjoyable even to the non-scientific reader. He will expel many false claims of the Fire Ant menace and enlighten the reader with facts gathered from over 30 years of observations and experiments.”
- “There are lovely essays on the behavior of ant researchers interspersed among the more numerous and scientifically dense chapters. It is really rather astonishing all that Tschinkel and his fellows have been able to ask the ants experimentally and get them to reply… Tschinkel's volume is a beautiful monument to fire ants and to science.
Ant Architecture: the Wonder, Beauty and Science of Underground Nests. Princeton University Press, 2021. This is an account of my invention of the research field of ant nest architecture. In it, I describe the many steps and inventions needed to make perfect casts of underground nests, and to perform exact and complete excavations of live nests. In a series of field experiments, I explore whether the ants have an architectural plan, how they know where they are underground, how they dig new nests and move into them and how nest architecture relates to the division of labor among the workers. Final chapters speculate about the evolution of ant nests as well as the future of the study of ant nest architecture.
