
Professor of Biological Science
Mailing address:
Dr. Betty J. Gaffney
3021 Country Club Loop NW
Olympia, WA 98502
Phone: 850-531-0693
gaffney@bio.fsu.edu
Phone: 850-531-0693
Professor of Biological Science
Mailing address:
Dr. Betty J. Gaffney
3021 Country Club Loop NW
Olympia, WA 98502
gaffney@bio.fsu.edu
Overview: Reading the biological lipid code
I have been fascinated with "reading the biological lipid code" throughout my career. That code encompasses physical properties of lipid arrays all the way to enzymes that use membrane components as substrates- different ways of rendering cell membranes dynamic. The goal is to find the messages in these dynamics. I contributed to chemical synthesis of nitroxide spin labels in the beginning and applied EPR (electron paramagnetic resonance) spectroscopy to reveal the lifetime of lipid conformations in synthetic lipid bilayers, during post-doc periods in K. Nakanishi's and in H.M. McConnell's labs [3, 4, 11].
In early studies of lipid membrane structure (1970s), it was unclear if real biological membranes and lipid model bilayers had similar properties. As a new faculty member, I showed that the abnormal appearance of transformed fibroblasts does NOT indicate a global change in lipid properties from their normal counterparts, as determined with spin labeled fatty acids [15]. In contrast, mobility of phospholipid spin labels in the close-packed membranes of Sindbis virus changes dramatically when the E1/E2 proteins are removed by proteolysis [13]. The existence of patches of membrane with specialized physical properties was foreshadowed by these early papers. I also pursued other studies of lipid dynamics, culminating in discovery (and naming) of the "sub-transition" that occurs between low temperature phases of saturated lipid bilayers [30] and in synthesis of spin-labeled, bifunctional crosslinking reagents [40, 41]. These interests broaden more recently to include the lipid remodeling enzyme lipoxygenase and new applications of biological EPR. Some results are highlighted below.
Structure and mechanism of lipoxygenases
Membrane components are constantly remodeled to signal the state of a cell and to maintain fluidity. In the early 1980s (pre-cloning), the lipid remodeling lipoxygenases were available in abundance only from soybeans and animal reticulocytes. I improved soybean lipoxygenase purification and initiated a project with crystallographers J. Boyington and L.M. Amzel. The inaugural structure of the enzyme [58, 66, 68] has become a prototype of the lipoxygenase fold (PF00305).
Two lipoxygenase types were known early on: 5- and 15-lipoxygenases, the numbers referring to the carbon bearing -OOH in products from arachidonic acid. Pathways to mediators of inflammation differ for the two products. We had solved a 15-LOX structure, and finding determinates of specificity has guided my continuing studies. Recently we determined the location and dynamics of a spin labeled lipid bound to lipoxygenase [100], using advanced pulsed EPR studies (at ACERT resource, Cornell) (Figure 1). I also contributed to structural studies by other labs interested in determinants of product stereochemistry [94], in lipoxygenase isoforms of soybean [95], in manganese lipoxygenase [88], and in a bacterial lipoxygenase-phospholipid complex [101].
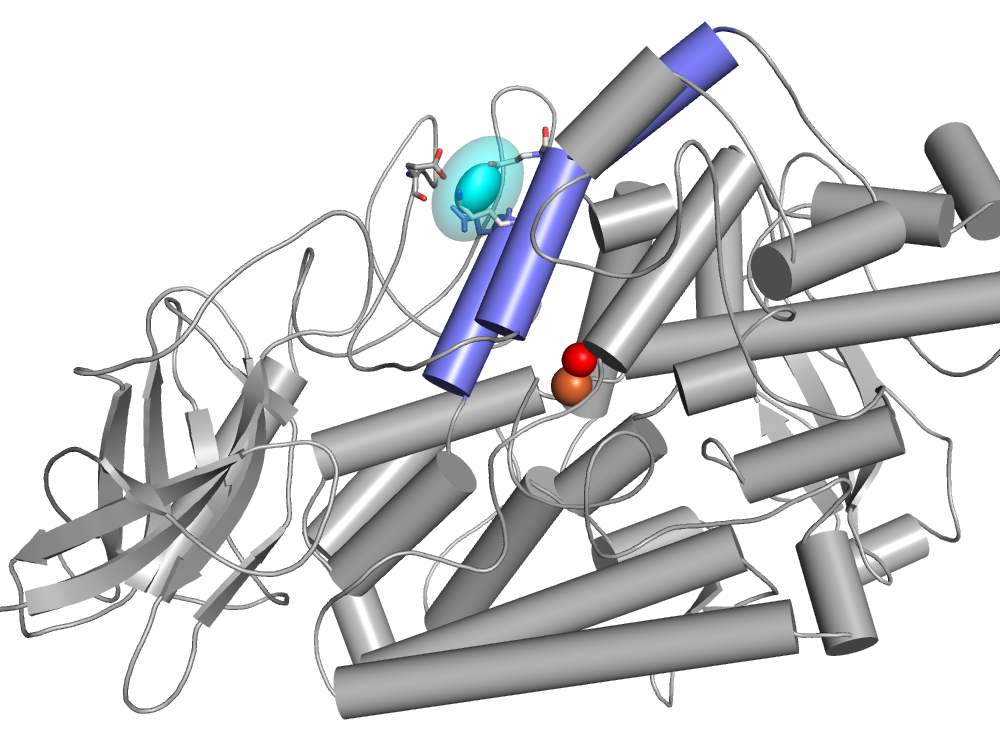
Fig. 1 Cyan ellipse: distribution of head group locations for a spin labeled lysolipid bound to soybean lipoxygenase. The lipid acyl chain must pass between the blue helices to approach the catalytic metal-water site. Iron center: rust, water: red.
The mutants of soybean lipoxygenase-1, prepared for spin label studies of structure and dynamics of the lipoxygenase [96. 100, 102], are deposited with Addgene.
Applications in Biological Magnetic Resonance
The EPR spectra of metal centers having more than one unpaired spin are intriguing and complex. I contributed to in depth reevaluation of spin 5/2 EPR spectra with quantum chemist, H. Silverstone [53]. Ferric transferrin is an historical model of non-heme iron in proteins. The mysterious EPR spectra of diferric transferrin carbonate was shown to arise from a distribution in zero field energies and the spectra could be simulated quantitatively [47]. This is confirmed in our more recent EPR measurements at the higher frequency, 94 GHz [80]. We applied similar simulation methods to studies of the non-heme iron enzymes phenylalanine hydroxylase [46] and lipoxygenases [54], showing, for instance, that only a single equivalent of lipid hydroperoxide is required to convert all the iron in resting ferrous P. aeruginosa lipoxygenase to the active ferric form [101]. We also employed higher frequency EPR as the basis for proposing that metal centers in iron and a manganese-containing lipoxygenase have similar ligand environments [88]. Further, we contributed to an NMR study in the R. Bowers lab, at UF, of hyperpolarized xenon interacting with proteins, including with lipoxygenase [84].
The complexity and beauty of metallo EPR spectra at X-band are illustrated in Figure 2, showing the many transitions occurring in a magnetic field scan of manganese lipoxygenase at 9.4 GHz [103]. Simplicity replaces beauty in a spectrum recorded at the higher frequency, 94 GHz (not shown) [88].
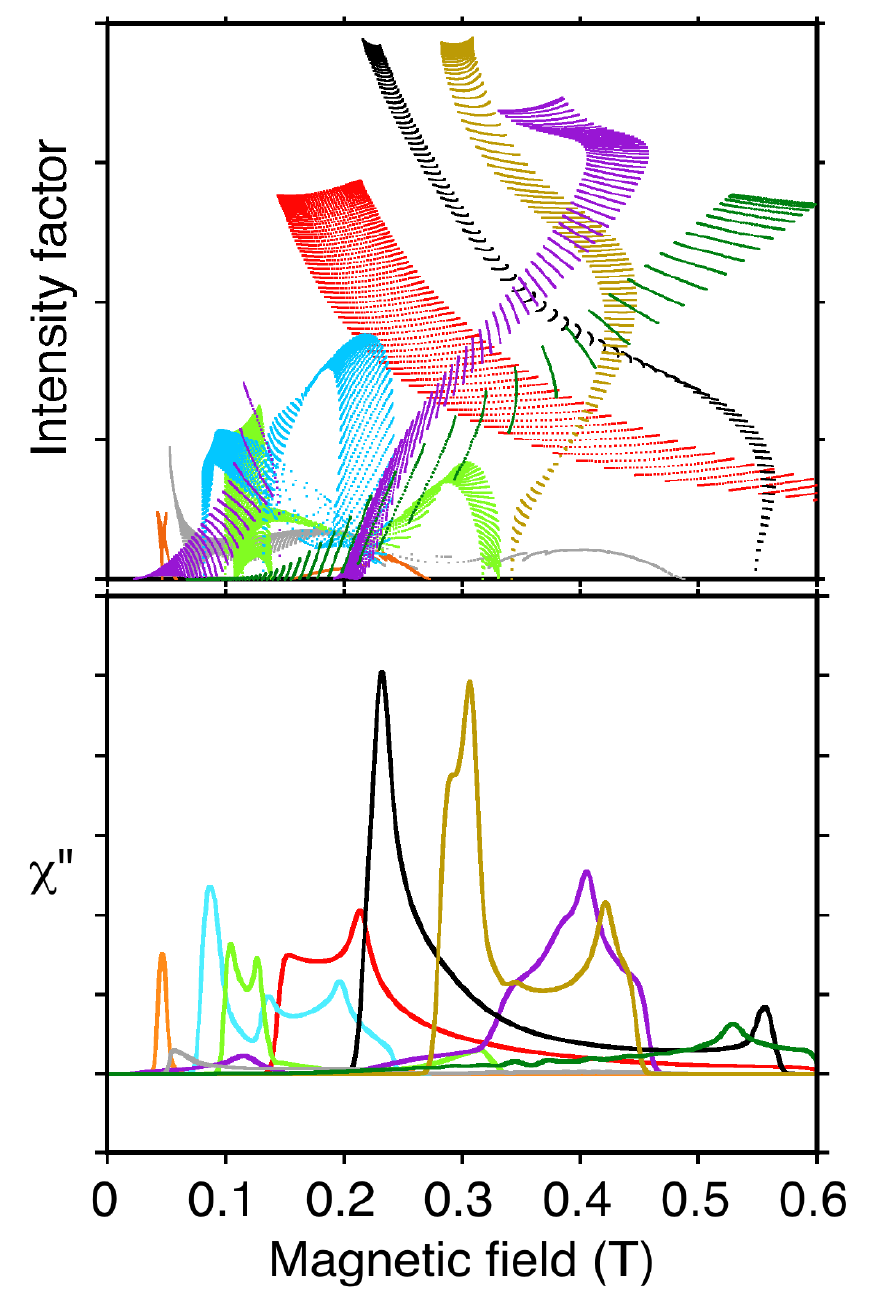
Fig. 2. Each dot (upper) represents EPR intensity from one molecular orientation in the magnetic field. These intensities sum for all orientations to give the components of observed spectra (lower). Transitions between different pairs of energy levels are represented by the various colors.
References: numbers refer to my publication list, updated in 2015, which can be found (here on my personal home page). A list of my publications can also be found at (with slightly different numbering) http://www.ncbi.nlm.nih.gov/sites/myncbi/betty.gaffney.1/bibliography/40434843/public/?sort=date&direction=descending